Using Neural Nets to analyse Li-metal battery quality
New research on using computer vision to identify dendrites in Lithium batteries
This post is an ELI5-style explanation of the paper Lithium Metal Battery Quality Control via Transformer-CNN Segmentation published by researchers at UC Berkeley, Lawrence Berkeley National Laboratory and UC Irvine.

Remember Facebook’s photo-tagging feature? You could tell it which of your friends were in a picture and after ‘tagging’ a friend in a few photos, the algorithm would be able to pick that friend out in more pictures. That feature is an application of Computer vision - a subset of AI that is able to determine the presence of patterns and/or objects in images once an underlying model has been trained on similar data.
The paper describes an experiment where a collection of X-ray images of Lithium-metal batteries are shown to a neural net. Some images are labeled as containing dendrites or not - think of tagging an image as `dendrites_present` or `dendrites_not_seen`. Most of the images are left without labels. After seeing the labeled and unlabeled images, the model is asked to determine whether a given X-ray image contains dendrites or not. Researchers find that the model is able to do this well enough to be useful for the task, in place of humans.
Why is anyone interested in identifying dendrites in lithium metal batteries?
In a Lithium-ion battery, Lithium ions (red circles in the picture below) travel from the cathode to anode during charging. During discharging, the same Lithium ions move in reverse - from the anode to the cathode. An important characteristic of a battery is this reversibility - the ability of ions to flow in one direction during charging and in reverse during discharging.
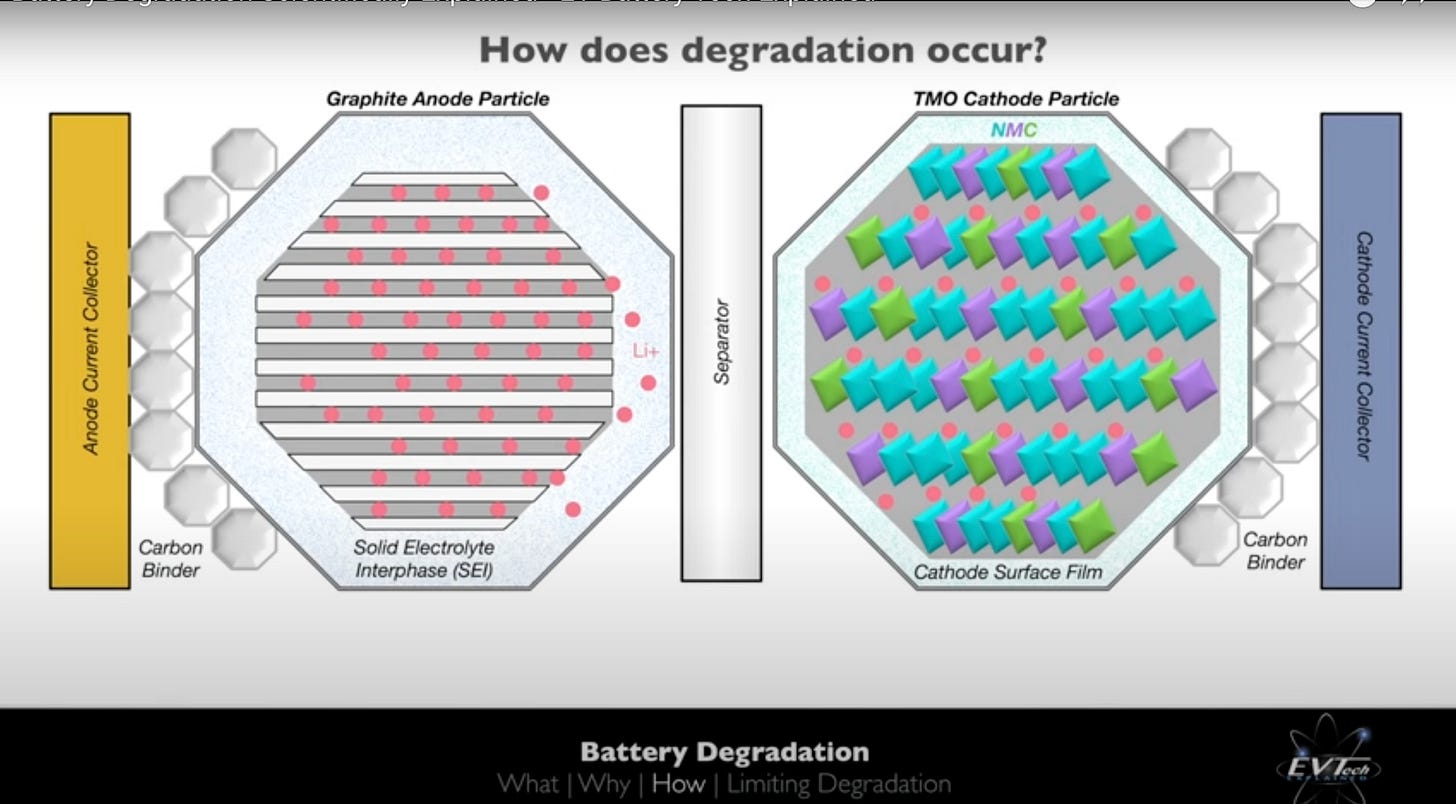
Currently, most Li-ion batteries use a Graphite anode and transition metals (such as Cobalt, Nickel, Manganese) in the cathode. There is a limit to the energy density - the energy per unit weight and volume - that can be achieved with this electrode combination. Using Lithium metal anodes, instead of graphite, can increase battery capacity by upto a factor of 10. This allows for nearly 10 times as much battery capacity at the same anode volume and weight.

But the formation of dendrites makes Lithium metal batteries less safe and susceptible to faster capacity loss.
Dendrites, also called whiskers, are tree-branch-like structures made of lithium that start forming on the anode surface during the charging-discharging process. They damage the battery in 2 ways:
Reduce battery capacity: Lithium that gets deposited on the anode surface will no longer move back and forth between the anode and cathode. Thus, in any given charge-discharge cycle, less electrical charge is transferred between the 2 electrodes resulting in lower battery capacity.
Cause safety issues: lithium ions in dendrites can react with the surrounding electrolyte in exothermic (heat-releasing) reactions greatly increasing internal battery temperatures and damaging battery components. Occasionally, if the dendrites (think of a thin metallic wire) grow long enough to penetrate the separator membranes and reach the cathode, an internal short circuit can also occur.
Here’s a video showing the formation of dendrites on the electrode surface inside a battery.
As we learnt earlier, during charging lithium ions flow from the cathode to anode and then enter the anode. If these two processes - flow, and entry into anode - happen at different rates, lithium ions can deposit on the surface of the anode instead of entering it. When a battery is charged at low temperature or when a fast charger is used, the ions enter the anode more slowly than the rate at which they flow from cathode. As a result, the lithium gets deposited on the surface of the anode rather than diffusing inside it. Over time, dendrites form and grow.
A specific kind of X-ray imaging has sufficient resolution to show Lithium dendrites inside a battery. But, under X-rays, dendrites appear similar to the ‘pits’ that develop on the battery’s electrodes during the charging and discharging process. The researchers train various neural nets to look at the X-ray images and classify them as showing dendrites or not. This helps battery researchers quickly discover areas of the battery where dendrites were seen by the neural net and focus on those areas to find out how dendrite formation can be prevented or slowed. Current X-ray imaging techniques generate large volumes of data and using a trained neural net can help battery researchers focus on images of interest. Another way AI is helping humans work smarter, faster and better!
P.S. If you use an Electric vehicle, EV-Tech Explained has some recommendations to make your battery last longer:
Don’t park in the sun
Avoid charging right after a long trip - wait for the battery to cool down
Avoid fully charging (above 90%) and fully discharging (less than 15%) the battery
Choose slow charging
I used Telborg’s CerebrumAI to do research for this post. CerebrumAI is an an AI research tool to help with your research on batteries and energy storage. Try it here.
I’m building Telborg and would love your feedback on CerebrumAI. Reach out on Twitter, LinkedIn or soumya@telborg.com.
References:
Article - A look inside your battery: Watching the dendrites grow
Paper - Understanding and preventing dendrite growth in Lithium metal batteries
Paper - Effects of lithium dendrites on thermal runaway and gassing of LiFePO4 batteries
Video - Scientists tackle the lithium dendrite problem in batteries
Video - Battery degradation scientifically explained, EV-Tech Explained
Video - New visualisation techniques to study Li-ion batteries, Michigan Engineering
Video - Taming 'dendrites' could bring better lithium-ion batteries, Purdue Engineering